ARA-290
Nerve Regeneration. Targeted Inflammation Control.
This peptide content is educational only and is not an offer to sell, prescribe, compound, or distribute any peptide or compounded product.
← Back to Peptide LibraryWhat It Treats

How It Works
ARA-290 selectively binds the innate repair receptor (IRR) — a heteromeric complex of the EPO receptor and CD131 — expressed on injured or stressed tissue. IRR activation drives tissue protection, reduces pro-inflammatory cytokines (TNF-α, IL-6), and promotes nerve fiber regeneration. Critically, ARA-290 does NOT bind the homodimer EPO receptor on red cell precursors, so it produces no hematocrit elevation.
Mechanism of Action
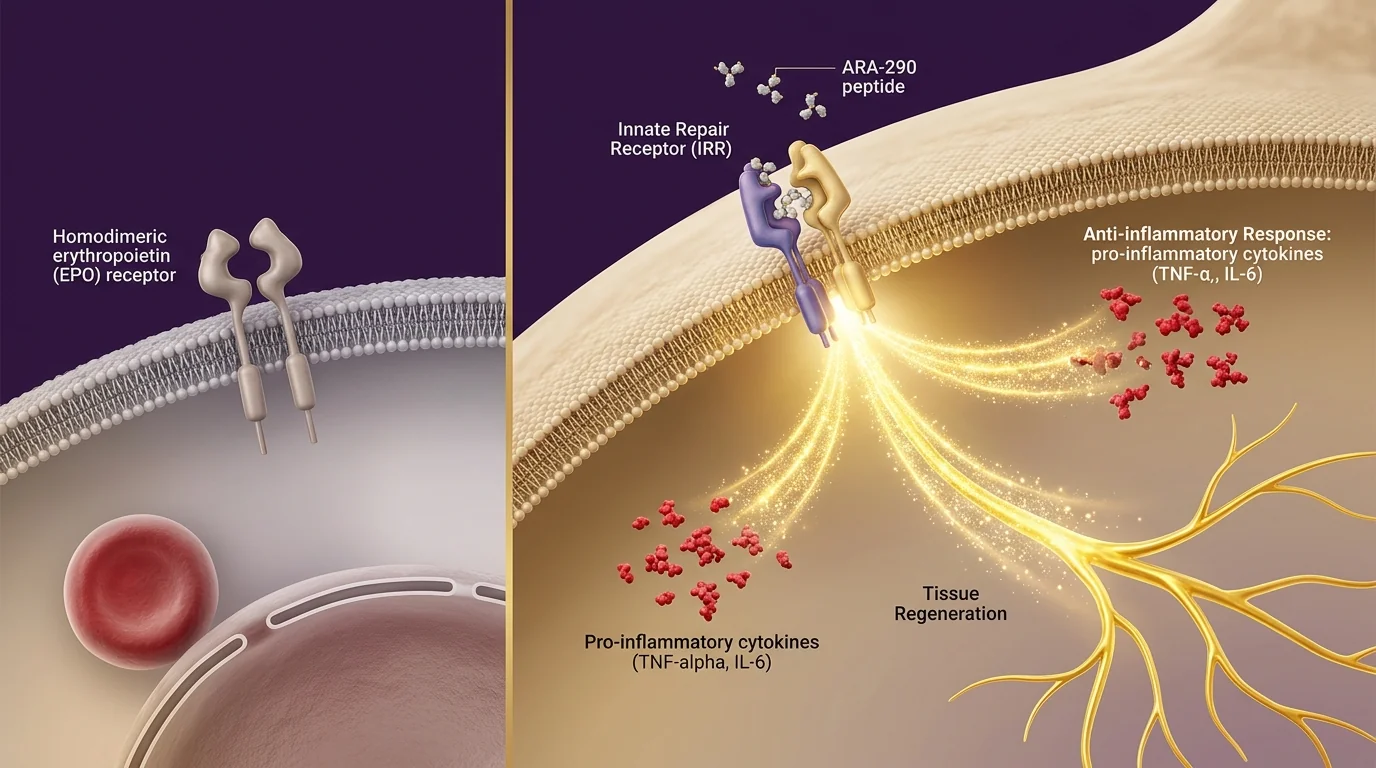
ARA-290 is an 11-amino-acid peptide derived from erythropoietin (EPO) — but with a critical difference: it was engineered to keep the *tissue-protective* signaling of EPO while eliminating the *blood-building* effect that causes polycythemia risk. Here is how that separation works at the receptor level. Full EPO binds a homodimer receptor on red blood cell precursors (causing hematocrit elevation) AND a heteromeric "innate repair receptor" (IRR) on injured or stressed tissue (causing protection and repair). ARA-290 binds *only* the IRR. Research published in Proceedings of the National Academy of Sciences (Brines et al.) first characterized this selectivity, and follow-up trials in Molecular Medicine demonstrated measurable improvements in neuropathic pain, nerve fiber density, and metabolic function in patients with small-fiber neuropathy — with no effect on hematocrit. This is why ARA-290 has emerged as a leading candidate for conditions where you want regeneration signaling without the cardiovascular risks of EPO itself.
In a Phase 2 trial of patients with small-fiber neuropathy, ARA-290 produced significant reductions in neuropathic pain (40% improvement vs placebo) and improvements in quality of life metrics. Importantly, intra-epidermal nerve fiber density — the gold-standard histological measure of small-fiber regeneration — increased measurably in the treatment group with no effect on hematocrit.
In sarcoidosis patients with documented small-fiber neuropathy, 28 days of ARA-290 produced significant improvements in neuropathic pain, autonomic symptoms, fatigue, and quality of life. The peptide also reduced levels of pro-inflammatory cytokines. Effects persisted beyond the treatment period, suggesting genuine tissue modification rather than symptomatic relief.
The Transformation
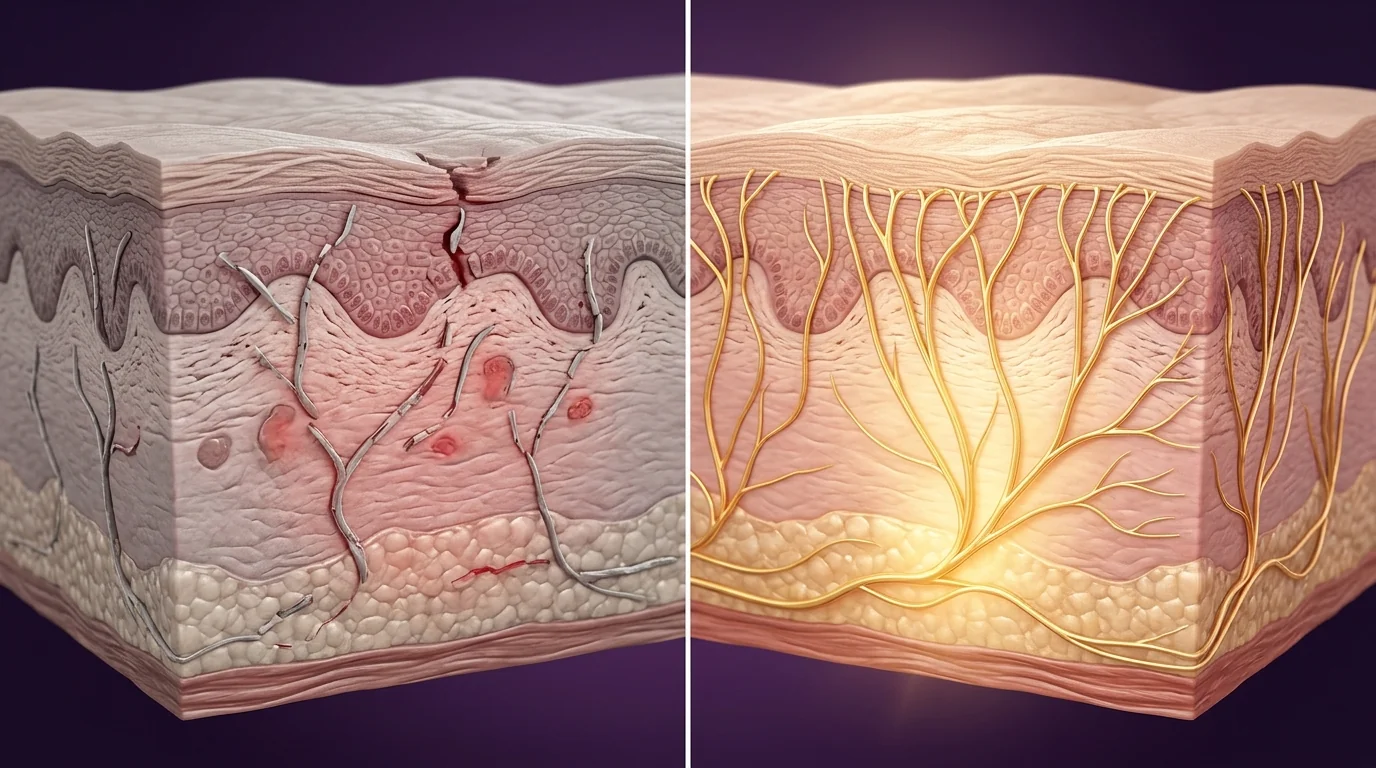
On the left: small-fiber neuropathy presenting as burning pain, numbness, reduced nerve fiber density in skin biopsy, and autonomic dysfunction — a pattern common in diabetic neuropathy, chemotherapy-induced neuropathy, and sarcoidosis-associated neuropathy. On the right, after a typical 12-week ARA-290 protocol: measurable improvements in intra-epidermal nerve fiber density, reduced neuropathic pain scores, and improved autonomic function. A Phase 2 trial in Molecular Medicine showed 40% reduction in pain scores and significant improvements in quality-of-life metrics. The distinction from pain medications: ARA-290 is not masking symptoms — it is driving genuine nerve fiber regeneration measured histologically.
Foundational study characterizing the innate repair receptor (IRR) as a distinct molecular target from the EPO receptor on red cell precursors. ARA-290 and related tissue-protective peptides bind IRR selectively, producing the regenerative and anti-inflammatory effects of full EPO without the cardiovascular risks of hematocrit elevation.
Double-blind placebo-controlled trial in sarcoidosis patients with small-fiber neuropathy confirmed 28-day ARA-290 treatment produced significant improvements in neuropathic pain scores, autonomic symptoms, fatigue, and quality of life — with benefits sustained beyond the treatment window. Hematocrit was unaffected, reinforcing the IRR-selective mechanism.
Comprehensive mechanistic review establishing ARA-290 and related IRR agonists as the first class of therapeutics that uncouple EPO's tissue-protective signaling from its erythropoietic activity. The authors outlined therapeutic potential across diabetic complications, neuropathic pain, cardiac ischemia-reperfusion injury, and sarcoidosis — with Phase 2 clinical validation for neuropathic indications.
What to Expect
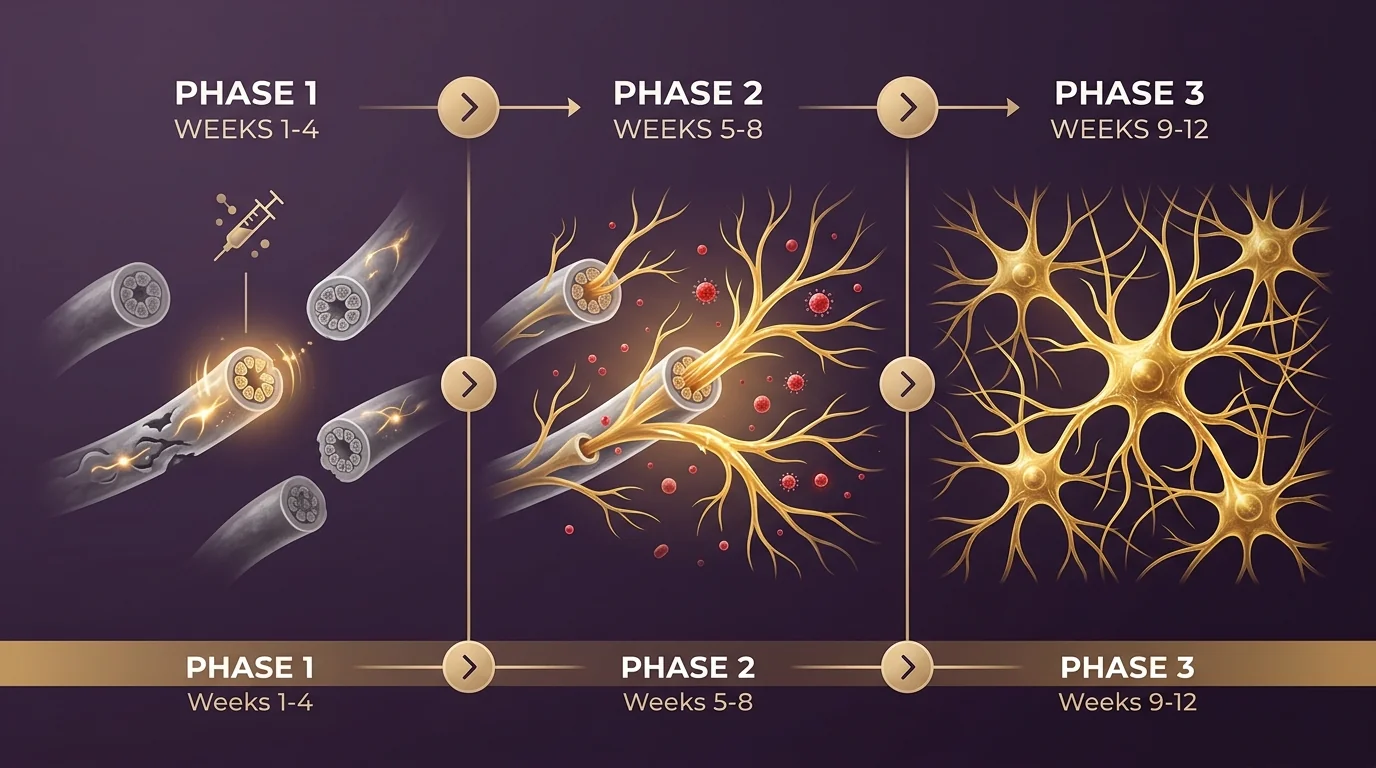
Initiation
Provider-directed home injection plan begins. Baseline labs and symptom assessment. Most patients notice early reductions in burning-pain intensity and improved sleep by week 3-4 as anti-inflammatory signaling takes hold.
Active Regeneration
Provider-directed adjustment may occur based on response. Intra-epidermal nerve fiber density begins measurable regeneration (confirmed histologically in clinical trials). Autonomic symptoms and neuropathic pain scores continue improving steadily.
Consolidation
Maintenance dosing. Sustained reductions in pain scores and inflammatory markers. Functional improvements in daily activities and sleep quality become stable. End-of-cycle labs and symptom reassessment.
Your Protocol at a Glance
Ideal For
Patients with documented small-fiber neuropathy (diabetic, chemotherapy-induced, or idiopathic), sarcoidosis patients with neuropathic symptoms, and those pursuing systemic anti-inflammatory protocols. Provider-supervised with lab monitoring.
Safety & Considerations
- Provider supervision required throughout the protocol with lab monitoring at baseline and end-of-cycle
- Contraindicated in patients with active malignancy without oncology clearance
- Should not be combined with erythropoietin-stimulating agents (ESAs) — disclose all current medications at intake
- Mild injection-site reactions reported in under 5% of patients; serious adverse events exceedingly rare in clinical trials
- Unlike full EPO, ARA-290 does not elevate hematocrit — this is the key safety differentiator that makes it suitable for patients with cardiovascular risk factors
Frequently Asked Questions
Educational Reference Only
This peptide content is educational only and is not an offer to sell, prescribe, compound, or distribute any peptide or compounded product.
This content is not an offer to sell, prescribe, compound, or distribute any medication, peptide, or compounded product.
Explore Other Peptides
 BPC-157
BPC-157 TB-500
TB-500 Tesamorelin
Tesamorelin Sermorelin
Sermorelin CJC-1295
CJC-1295 Semax
Semax Copper Peptide (GHK-Cu)
Copper Peptide (GHK-Cu) PT-141 (Bremelanotide)
PT-141 (Bremelanotide) Melanotan
Melanotan MOTS-c
MOTS-c Glutathione Injections
Glutathione Injections KPV
KPV DSIP
DSIP High Dose Vitamin C
High Dose Vitamin C Venofer (Iron Infusion)
Venofer (Iron Infusion) Wolverine (Recovery Stack)
Wolverine (Recovery Stack) Captain America (Sleep Stack)
Captain America (Sleep Stack) Black Widow (Glow Blend)
Black Widow (Glow Blend) Deadpool (Healing+ Stack)
Deadpool (Healing+ Stack)